The Lever Rule is a simple graphical relationship between the proportions of phases in a mixture. If you have a good understanding of the Lever Rule, you will have a powerful tool at your disposal for the study of igneous and metamorphic rocks. Because the weight percent proportions must add up to 100, when there are only two phases (such as brine and halite) there is only one variable.
Figure 3.05. Chemical composition diagram. The proportions of saturated brine and halite mixtures to the right of the dashed line can be read from the green composition axis. Click on the graph to see a larger, interactive version with more information about the green composition axis and the lever rule.
As you saw in the enlarged version of Figure 3.05, the bulk composition of a mixture of two phases divides the line between them (a tie line) into two parts whose relative lengths give the relative masses of the two phases. This relationship is called the
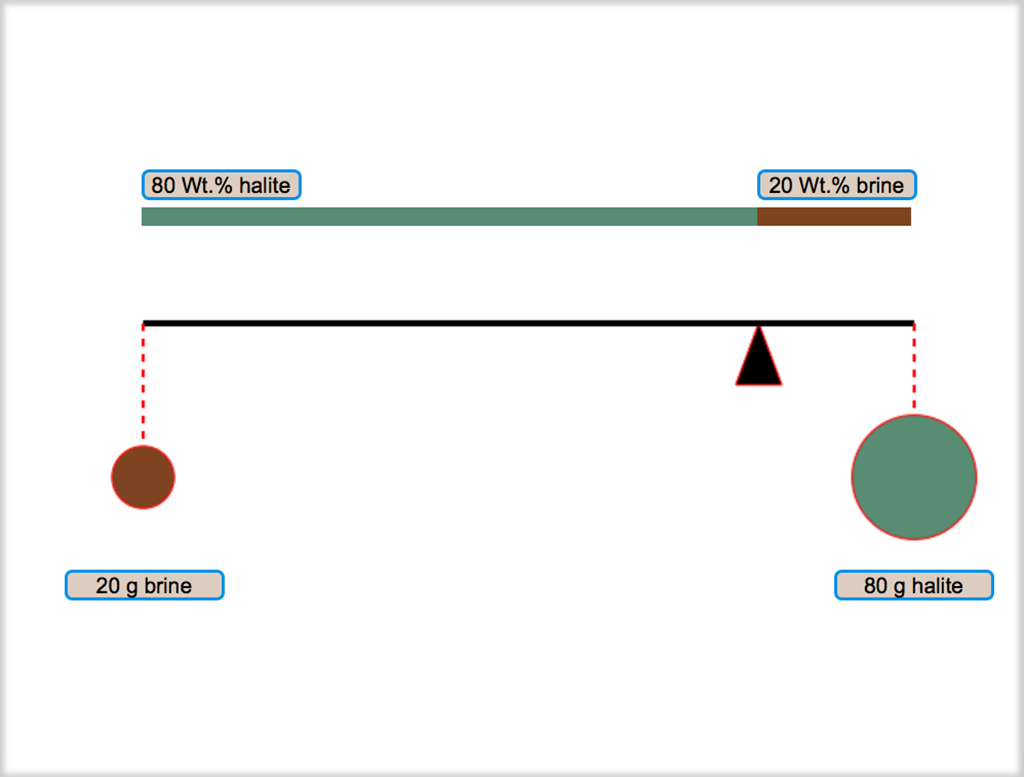
Figure 3.06. Lever Rule with weights. Click on the image for a larger version and to learn more about the lever rule.
Press "Enter" after you type in the number.
Excellent! You show good understanding of the Lever Rule.
Press "Enter" after you type in the number.
Excellent! Now you are making sense of the the Lever Rule.
Use the graphical tools and try again.
Press "Enter" after you type in the number.
Excellent! Now you are making sense of the the Lever Rule.
- The brine has all the water (50g of H2O).
- The brine is saturated with halite and must, therefore, be 25 weight percent NaCl and 75 weight percent H2O.
- 75 weight percent of the brine is all the H2O (50g).
- The 25 weight percent of the brine that is NaCl must be 50g*(25/75) = 16.67g.
- The mass of the brine is 50g (H2O) + 16.67g (NaCl) = 66.67g
- The remaining NaCl (50g - 16.67g = 33.33g) must be in halite.
- The weight percent of brine in the mixture is 100*[66.67g/(66.67g + 33.33g)] = 66.67%.
You can check this result using the enlarged version of Figure 3.05. Click on the Coordinates button to locate the bulk composition (50 weight percent H2O and 50 weight percent NaCl. Click on the Lever Rule button. Drag the Lever Rule handle to match that bulk composition and you will be able to read the weight percent brine in the mixture.